An antimicrobial peptide that inhibits translation by trapping release factors on the ribosome
24-Jul-2017
Nature Structural & Molecular Biology, volume 24, 752–757, doi:10.1038/nsmb.3439
Nature Structural & Molecular Biology, online article
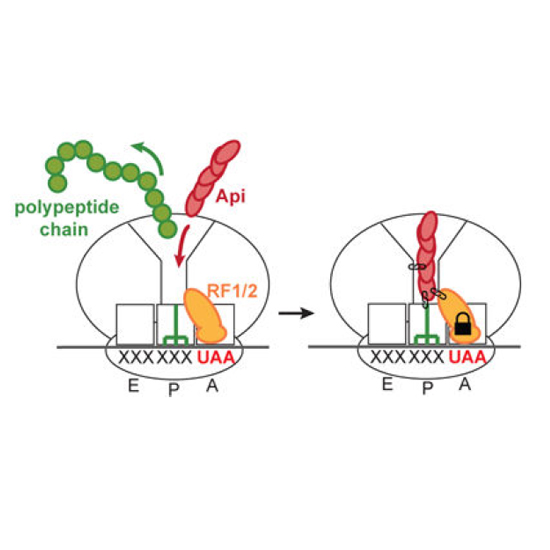
Many antibiotics stop bacterial growth by inhibiting different steps of protein synthesis. However, no specific inhibitors of translation termination are known. Proline-rich antimicrobial peptides, a component of the antibacterial defense system of multicellular organisms, interfere with bacterial growth by inhibiting translation. Here we show that Api137, a derivative of the insect-produced antimicrobial peptide apidaecin, arrests terminating ribosomes using a unique mechanism of action. Api137 binds to the Escherichia coli ribosome and traps release factor (RF) RF1 or RF2 subsequent to the release of the nascent polypeptide chain. A high-resolution cryo-EM structure of the ribosome complexed with RF1 and Api137 reveals the molecular interactions that lead to RF trapping. Api137-mediated depletion of the cellular pool of free release factors causes the majority of ribosomes to stall at stop codons before polypeptide release, thereby resulting in a global shutdown of translation termination.