Calcium depletion and calmodulin inhibition affect the import of nuclear-encoded proteins into plant mitochondria
19-Feb-2009
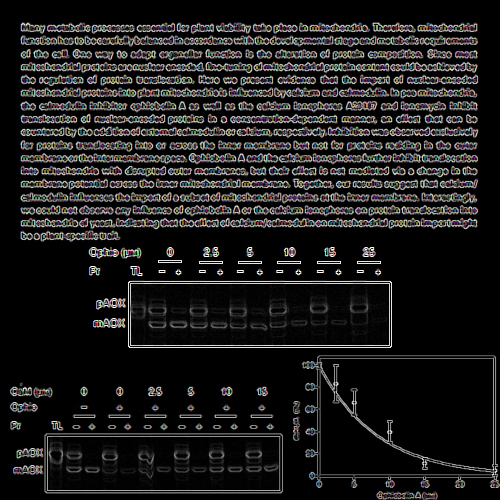
Many metabolic processes essential for plant viability take place in mitochondria. Therefore, mitochondrial function has to be carefully balanced in accordance with the developmental stage and metabolic requirements of the cell. One way to adapt organellar function is the alteration of protein composition. Since most mitochondrial proteins are nuclear encoded, fine-tuning of mitochondrial protein content could be achieved by the regulation of protein translocation. Here we present evidence that the import of nuclear-encoded mitochondrial proteins into plant mitochondria is influenced by calcium and calmodulin. In pea mitochondria, the calmodulin inhibitor ophiobolin A as well as the calcium ionophores A23187 and ionomycin inhibit translocation of nuclear-encoded proteins in a concentration-dependent manner, an effect that can be countered by the addition of external calmodulin or calcium, respectively. Inhibition was observed exclusively for proteins translocating into or across the inner membrane but not for proteins residing in the outer membrane or the intermembrane space. Ophiobolin A and the calcium ionophores further inhibit translocation into mitochondria with disrupted outer membranes, but their effect is not mediated via a change in the membrane potential across the inner mitochondrial membrane. Together, our results suggest that calcium/ calmodulin influences the import of a subset of mitochondrial proteins at the inner membrane. Interestingly, we could not observe any influence of ophiobolin A or the calcium ionophores on protein translocation into mitochondria of yeast, indicating that the effect of calcium/calmodulin on mitochondrial protein import might be a plant-specific trait.