Coupled chaperone action in folding and assembly of hexadecameric Rubisco
14-Jan-2010
Nature, 2010, doi:10.1038/nature08651, Volume 463, 197-202 published on 14.01.2010
Nature, online article
Nature, online article
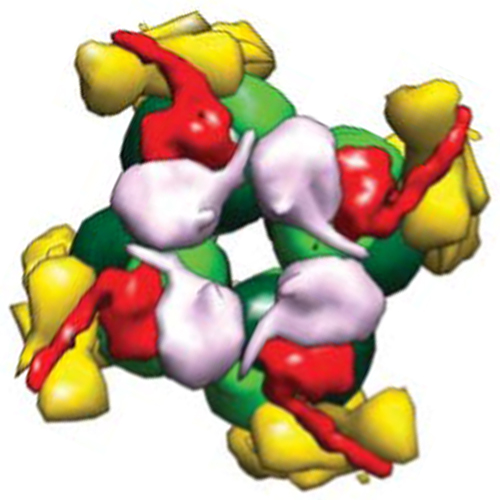
Form I Rubisco (ribulose 1,5-bisphosphate carboxylase/oxygenase), a complex of eight large (RbcL) and eight small (RbcS) subunits, catalyses the fixation of atmospheric CO2 in photosynthesis. The limited catalytic efficiency of Rubisco has sparked extensive efforts to re-engineer the enzyme with the goal of enhancing agricultural productivity. To facilitate such efforts we analysed the formation of cyanobacterial form I Rubisco by in vitro reconstitution and cryo-electron microscopy. We show that RbcL subunit folding by the GroEL/GroES chaperonin is tightly coupled with assembly mediated by the chaperone RbcX2. RbcL monomers remain partially unstable and retain high affinity for GroEL until captured by RbcX2. As revealed by the structure of a RbcL8–(RbcX2)8 assembly intermediate, RbcX2 acts as a molecular staple in stabilizing the RbcL subunits as dimers and facilitates RbcL8 core assembly. Finally, addition of RbcS results in RbcX2 release and holoenzyme formation. Specific assembly chaperones may be required more generally in the formation of complex oligomeric structures when folding is closely coupled to assembly.