Locking GTPases covalently in their functional states
16-Jul-2015
Nature Communications, 2015, doi:10.1038/ncomms8773, 6, Article number: 7773 published on 16.07.2015
Nature Communications, online article
Nature Communications, online article
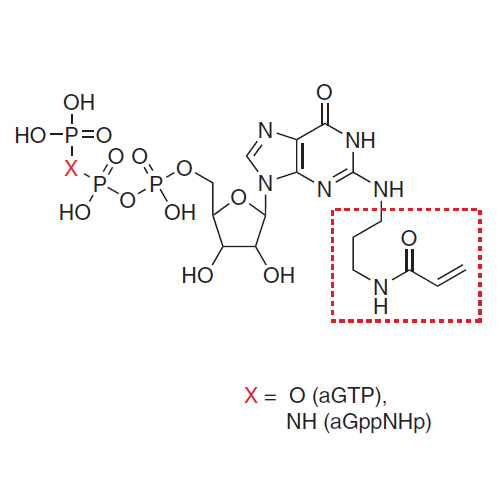
GTPases act as key regulators of many cellular processes by switching between active (GTP-bound) and inactive (GDP-bound) states. In many cases, understanding their mode of action has been aided by artificially stabilizing one of these states either by designing mutant proteins or by complexation with non-hydrolysable GTP analogues. Because of inherent disadvantages in these approaches, we have developed acryl-bearing GTP and GDP derivatives that can be covalently linked with strategically placed cysteines within the GTPase of interest. Binding studies with GTPase-interacting proteins and X-ray crystallography analysis demonstrate that the molecular properties of the covalent GTPase–acryl–nucleotide adducts are a faithful reflection of those of the corresponding native states and are advantageously permanently locked in a defined nucleotide (that is active or inactive) state. In a first application, in vivo experiments using covalently locked Rab5 variants provide new insights into the mechanism of correct intracellular localization of Rab proteins.