Monitoring Protein Conformation along the Pathway of Chaperonin-Assisted Folding
04-Apr-2008
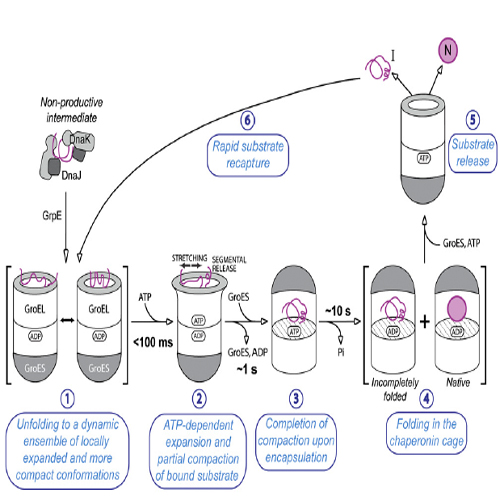
The GroEL/GroES chaperonin system mediates protein folding in the bacterial cytosol. Newly synthesized proteins reach GroEL via transfer from upstream chaperones such as DnaK/DnaJ (Hsp70).Here we employed single molecule and ensemble FRET to monitor the conformational transitions of amodel substrate as it proceeds along this chaperone pathway. We find that DnaK/DnaJ stabilizes the protein in collapsed states that fold exceedingly slowly. Transfer to GroEL results in unfolding,with a fraction ofmolecules reaching locally highly expanded conformations. ATP-induced domain movements in GroEL cause transient further unfolding andrapidmobilizationofprotein segments with moderate hydrophobicity, allowing partial compaction on the GroEL surface. The more hydrophobic regions are released upon subsequent protein encapsulation in the central GroEL cavity by GroES, completing compaction and allowing rapid folding. Segmental chain release and compaction may be important in avoiding misfolding by proteins that fail to fold efficiently through spontaneous hydrophobic collapse.