Light-triggered beta-hairpin folding and unfolding
02-Oct-2007
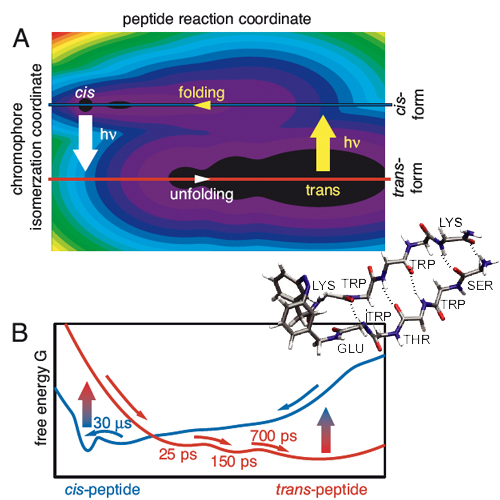
A light-switchable peptide is transformed with ultrashort pulses from aß-hairpin to an unfolded hydrophobic cluster and vice versa. The structural changes are monitored by mid-IR probing. Instantaneous normal mode analysis with a Hamiltonian combining density functional theory with molecular mechanics is used to interpret the absorption transients. Illumination of the b-hairpin state triggers an unfolding reaction that visits several intermediates and reaches the unfolded state within a few nanoseconds. In this unfolding reaction to the equilibrium hydrophobic cluster conformation, the system does not meet significant barriers on the free-energy surface. The reverse folding process takes much longer because it occurs on the time scale of 30 micro s The folded state has a defined structure, and its formation requires an extended search for the correct hydrogen-bond pattern of the ß-strand.