Mutations of the peripheral antenna complex LH2–correlations of energy transfer time with other functional properties
23-Feb-2009
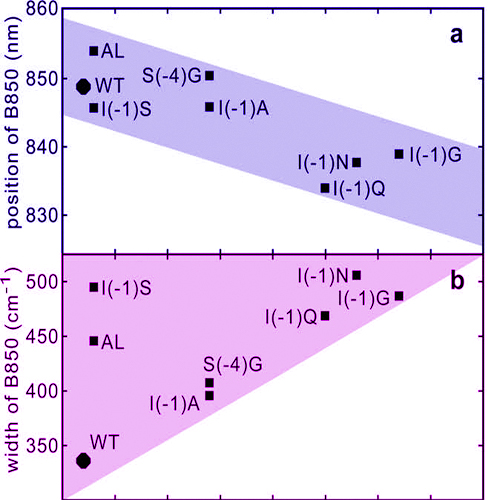
The peripheral antenna complex (LH2) of wild type and mutants of the LH2 α-subunit of purple bacterium Rhodobacter sphaeroides are investigated by transient and stationary absorption spectroscopy. The time for energy transfer from the bacteriochlorophyll (BChl) molecules B800 to B850 is found to depend on the mutation and varies between 0.7 ps for wild type LH2 to 1.0 ps. Also quantities of functional properties (peak position, width and relative band intensity) of the B850 absorption band depend on mutation. In correlation plots connections between different quantities and the energy transfer time are identified. The observed correlations indicate that the mutations influence the binding affinities of the BChl molecules. Heterogeneity in structure and changes in the occupation of B800 and B850 rings lead to the observed variation of functional properties.