- Home ·
- Publications ·
- Research Area A ·
- 2008 ·
Photophysics of New Water-Soluble Terrylenediimide Derivatives and Applications in Biology
15-Dec-2008
Christophe Jung, Nadia Ruthardt, Robert Lewis, Jens Michaelis, Beate Sodeik, Fabian Nolde, Kalina Peneva, Klaus Müllen, Christoph Bräuchle
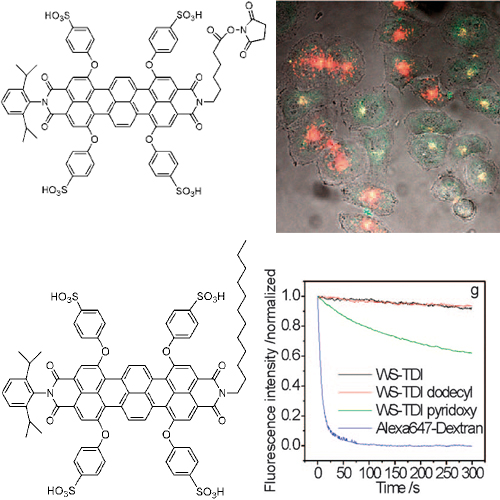
The photophysical properties of three new water-soluble terrylenediimide (WS-TDI) derivatives are investigated and their utilization in biological experiments is demonstrated. Each of these dyes can be excited in the far red region of the visible spectrum, making them good candidates for in-vivo studies. Single-molecule techniques characterize their photophysics that is, the number of emitted photons, blinking characteristics and survival times until photobleaching takes place. All three dyes exhibit bright fluorescence, as well as an extremely high resistance against photodegradation compared to other well-known fluorophores. Due to their different characteristics the three new WSTDI derivatives are suitable for specialized biological applications. WS-TDI dodecyl forms non-fluorescent aggregates in water which can be disrupted in a hydrophobic environment leading to a monomeric fluorescent form. Due to its high lipophilicity WS-TDI dodecyl anchors efficiently in lipid bilayers with its alkyl chain and hence can be ideally used to image membranes and membrane-containing compartments in living cells. In contrast, the positively charged WS-TDI pyridoxy is a new type of chromophore in the WS-TDI family. It is fully solubilized in water forming fluorescent monomers and is successfully used to label the envelope of herpes simplex viruses. Finally, it is shown that a WS-TDI derivative functionalized with N-hydroxysuccinimide ester moiety (WS-TDI/NHS ester) provides a versatile reactive dye molecule for the specific labelling of amino groups in biomolecules such as DNA.