Site-specific labelling of native mammalian proteins for singlemolecule FRET measurements
02-Feb-2018
ChemBioChem, 9(8) (2018, 780–783, DOI: 10.1002/cbic.201700696
ChemBioChem, online article
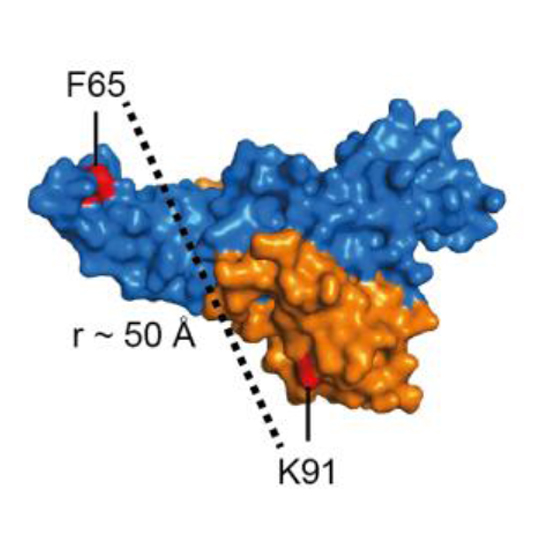
Human cells are complex entities in which molecular recognition and selection are critical for cellular processes often driven by structural changes and dynamic interactions. Biomolecules appear in different chemical states and modifications like phosphorylation affect their function. Hence, using proteins in their chemically native state in biochemical and biophysical assays is essential. Single-molecule FRET measurements allow the exploration of the structure, function and dynamics of biomolecules but cannot be fully exploited for the human proteome as a method for the site-specific coupling of organic dyes into native, non-recombinant mammalian proteins is lacking. We address this issue showing the site-specific engineering of fluorescent dyes into human proteins based on bioorthogonal reactions. We show the applicability of the method to study functional and posttranslationally modified proteins on the single-molecule level, among them the hitherto inaccessible human Argonaute 2.