A Distinct Mode of Interaction of a Novel Ketolide Antibiotic That Displays Enhanced Antimicrobial Activity
21-Jan-2009
Antimicrob. Agents Ch., 2009, 53(4), 1411-1419, doi:10.1128/AAC.01425-08 published on 21.01.2009
Antimicrob. Agents Ch., online article
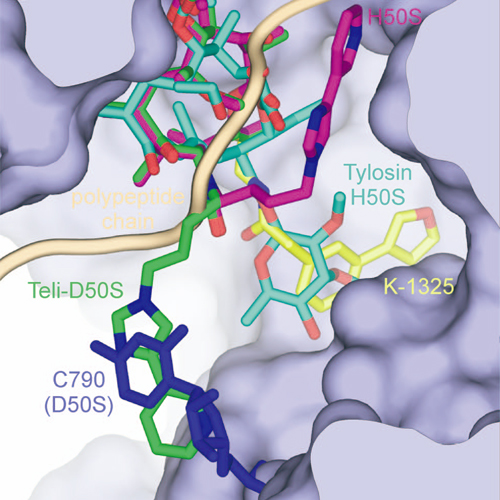
Ketolides represent the latest generation of macrolide antibiotics, displaying improved activities against some erythromycin resistant strains, while maintaining their activity against erythromycin susceptible ones. In this study we present a new ketolide K-1325 that carries an alkyl-aryl side chain at C-13 of the lactone ring. According to our genetic and biochemical studies K-1325 binds within the nascent polypeptide exit tunnel, at a site previously described as the primary attachment site of all macrolide antibiotics. Compared with telithromycin, K-1325 displays enhanced antimicrobial activity against wild type E.coli strains, as well as against strains bearing the U2609C mutation in 23S rRNA. Chemical protection experiments showed that the aryl-alkyl side chain of K-1325 interacts specifically with helix 35 of 23S rRNA, a fact leading to an increase affinity of U2609C mutant ribosomes for the drug, and rationalizing the enhanced effectiveness of this new ketolide.