A two-component adhesive: Tau fibrils arise from a combination of a well-defined motif and conformationally flexible interactions
26-Jan-2017
J. Am. Chem. Soc., Just Accepted Manuscript, DOI: 10.1021/jacs.6b09619
J. Am. Chem. Soc., online article
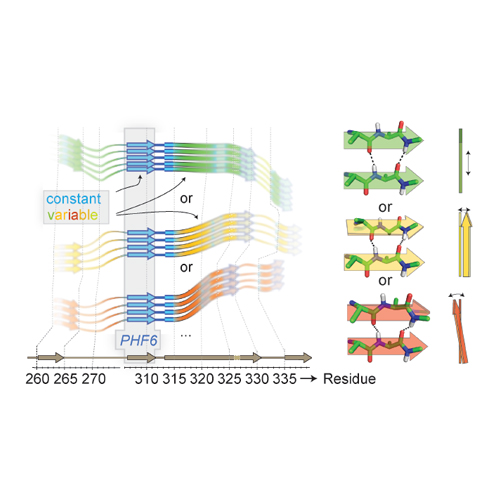
Fibrillar aggregates of Aβ and Tau in the brain are the major hallmarks of Alzheimer’s disease. Most Tau fibers have a twisted appearance but the twist can be variable and even absent. This ambiguity, which has also been associated with different phenotypes of tauopathies, has led to controversial assumptions about fibril constitution, and it is unclear to-date what the molecular causes of this polymorphism are. To tackle this question, we used solid-state NMR strategies providing assignments of non-seeded three-repeat-domain Tau3RD with an inherent heterogeneity. This is in contrast to the general approach to characterize the most homogeneous preparations by construct truncation or intricate seeding protocols. Here, carbon and nitrogen chemical-shift conservation between fibrils revealed invariable secondary-structure properties, however, with inter-monomer interactions variable among samples. Residues with variable amide shifts are localized mostly to N- and C-terminal regions within the rigid beta structure in the repeat region of Tau3RD. By contrast, the hexapeptide motif in repeat R3, a crucial motif for fibril formation, shows strikingly low variability of all NMR parameters: Starting as a nucleation site for monomer-monomer contacts, this 6-residue sequence element also turns into a well-defined structural element upon fibril formation. Given the absence of external causes in-vitro, the interplay of structurally differently conserved elements in this protein likely reflects an intrinsic property of Tau fibrils.