Amythiamicin D and Related Thiopeptides as Inhibitors of the Bacterial Elongation Factor EF-Tu: Modification of the Amino Acid at Carbon Atom C2 of Ring C Dramatically Influences Activity
17-Sep-2013
ChemMedChem, 2013, DOI: 10.1002/cmdc.201300323, Volume 8, Issue 12, pages 1954–1962 published on 17.09.2013
ChemMedChem, online aticle
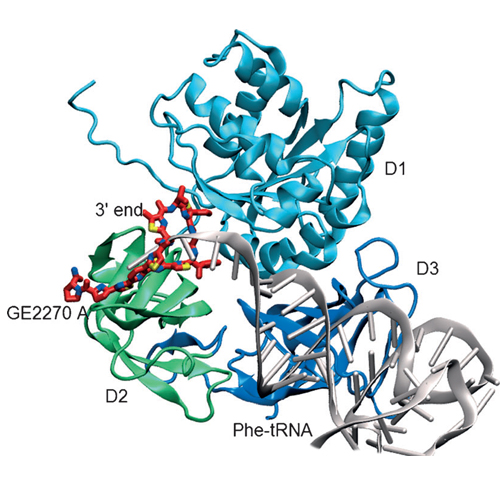
Three analogues of amythiamicin D, which differ in the substitution pattern at the methine group adjacent to C2 of the thiazole ring C, were prepared by de novo total synthesis. In amythiamicin D, this carbon atom is (S)-isopropyl substituted. Two of the new analogues carry a hydroxymethyl in place of the isopropyl group, one at an S- (compound 3 a) and the other at an R-configured stereogenic center (3 b). The third analogue, 3 c, contains a benzyloxymethyl group at an S-configured stereogenic center. Compounds 3 b and 3 c showed no inhibitory effect toward various bacterial strains, nor did they influence the translation of firefly luciferase. In stark contrast, compound 3 a inhibited the growth of Gram-positive bacteria Staphylococcus aureus (strains NCTC and Mu50) and Listeria monocytogenes EGD. In the firefly luciferase assay it proved more potent than amythiamicin D, and rescue experiments provided evidence that translation inhibition is due to binding to the bacterial elongation factor Tu (EF-Tu). The results were rationalized by structural investigations and by molecular dynamics simulations of the free compounds in solution and bound to the EF-Tu binding site. The low affinity of compound 3 b was attributed to the absence of a critical hydrogen bond, which stabilizes the conformation required for binding to EF-Tu. Compound 3 c was shown not to comply with the binding properties of the binding site.