An Asymmetric Dimer as the Basic Subunit in Alzheimer’s Disease Amyloid β Fibrils
08-May-2012
Angewandte Chemie, 2012, DOI: 10.1002/anie.201200965, Volume 51, Issue 25, pages 6136–6139, published on 08.05.2012
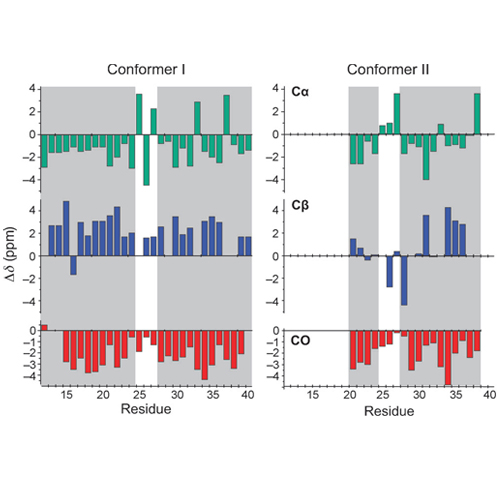
Aggregation of monomeric amyloid beta peptides (ABeta) into soluble oligomers and insoluble fibrils is one of the major pathological hallmarks of Alzheimer_s disease (AD). In the past few years, magic angle spinning (MAS) solid-state NMR spectroscopy has enabled considerable progress in the structural characterization of amyloid aggregates, and a number of structural models have been suggested. Amyloid samples fibrilize in a large variety of morphologies. Differences in fibril morphology have been attributed to differences in peptide conformation. These variations in the molecular structures of Abeta fibrils might be responsible for the variations in the toxicity and deposition patterns of fibrils in AD.[5] Whereas previous NMR studies implied that in all cases a U-shaped peptide fold forms the fundamental structural unit of the fibril, cryo electron microscopy studies suggested that different Abeta fibril morphologies can be constructed from a dimeric arrangement of two conformationally distinct peptide molecules in each molecular layer of the protofilament. To date, however, there are no solid-state NMR data that supports the existence of such a structural subunit in Abeta fibrils.