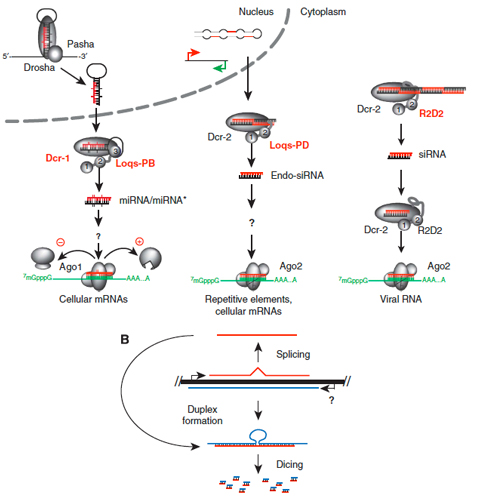
Endo-siRNAs depend on a new isoform of loquacious and target artificially introduced, high-copy sequences
04-Aug-2009
Transposons are mobile genetic elements found in the hereditary material of humans and other organisms. They can replicate and the new copies can insert at novel sites in the genome. Because this threatens the whole organism, molecular mechanisms have evolved which can repress transposon activity. CIPSM-Professor Klaus Förstemann of the Gene Center of Ludwig-Maximilians-Universität (LMU) in Munich and a team of researchers working with the fruitfly Drosophila melanogaster have now uncovered a new type of cellular defence that acts against DNA sequences present in high copy numbers inside the cell, even if they have not integrated into the genome. Small molecules of RNA (a class of nucleic acid closely related to the genetic material DNA) play the central role. “Transposons are genomic parasites, so to speak”, says Förstemann. “If they are allowed to proliferate, the genome can become unstable or cancers can develop. We now want to find out whether mammalian cells possess this newly discovered defence mechanism and to elucidate precisely how it works. Press Spiegel online Medical News Today Science Daily Transposons constitute a significant fraction of the genomes of most higher organisms. Indeed, it is estimated that these mobile elements, which include one or more genes, make up as much as half of the genetic material. “This demonstrates”, says Förstemann, “that it is not always possible to tame these “selfish” genetic elements, although highly efficient mechanisms of defence have evolved. For instance, in the germ cells, which are required for reproduction, the system of so-called piRNAs ensures that transposon activity is inhibited – but only if these RNAs are transmitted from the mother. Disruption of this system usually leads to a drastic reduction in the fertility of the progeny. Germ cells are an ideal target for transposons, since these cells pass their genetic material – together with integrated mobile elements – on to all cells of the progeny. But normal body (somatic) cells can also be attacked by transposons. For example, certain viruses carry transposons in their genomes and introduce them into the host cells they infect. Therefore, transposon activity must also be repressed in somatic cells. Recently so-called endo-siRNAs that perform this function were discovered in the fruitfly. A similar class of molecules has been found in mice. By means of a process called RNA interference, the siRNAs enable the cell to recognize and destroy messenger RNAs derived from transposons. The researchers in Förstemann’s group were able to identify a protein that is essential for the production of endo-siRNAs. It turns out that this is a previously unknown variant of the protein “Loquacious”. In Drosophila, Loquacious can bind to specific RNA molecules that serve as precursors of the endo-siRNAs. Furthermore, the team pinpointed an entirely novel feature of this system: Repression of transposon activity was also detectable when multiple copies of a mobile element were present in the cell but not yet incorporated into the genome. The phenomenon of RNA interference first came to light only a short time ago, but has already become a well established field of study. Thanks to more recent findings, the known repertoire of small RNAs has grown. As Förstemann stresses, “It is therefore particularly important to discriminate between the various molecular classes in terms of their modes of synthesis and specific functions”. This is no easy task, since all these molecules are similar in size and virtually indistinguishable chemically. “We will now test whether the mechanism we have found in drosophila also exists in mammalian cells. We would also like to know how the mechanism is targeted specifically against sequences present in high copy numbers”.