hnRNP A1 Proofreads 30 Splice Site Recognition by U2AF
10-Feb-2012
Molecular Cell, 2012, 10.1016/j.molcel.2011.11.033, Volume 45, Issue 3, 314-329 published on 10.02.2012
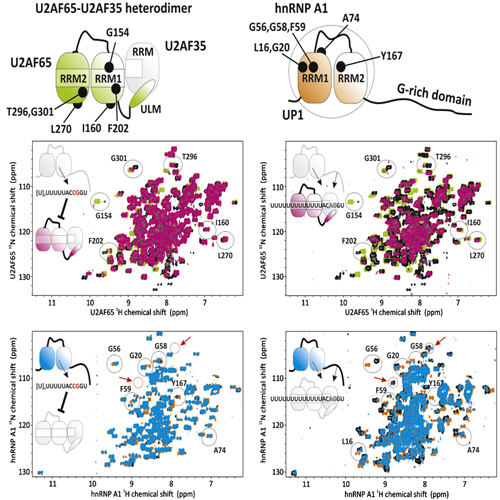
One of the earliest steps in metazoan pre-mRNA splicing involves binding of U2 snRNP auxiliary factor (U2AF) 65 KDa subunit to the polypyrimidine (Py) tract and of the 35 KDa subunit to the invariant AG dinucleotide at the intron 3′ end. Here we use in vitro and in vivo depletion, as well as reconstitution assays using purified components, to identify hnRNP A1 as an RNA binding protein that allows U2AF to discriminate between pyrimidine-rich RNA sequences followed or not by a 3′ splice site AG. Biochemical and NMR data indicate that hnRNP A1 forms a ternary complex with the U2AF heterodimer on AG-containing/uridine-rich RNAs, while it displaces U2AF from non-AG-containing/uridine-rich RNAs, an activity that requires the glycine-rich domain of hnRNP A1. Consistent with the functional relevance of this activity for splicing, proofreading assays reveal a role for hnRNP A1 in U2AF-mediated recruitment of U2 snRNP to the pre-mRNA.