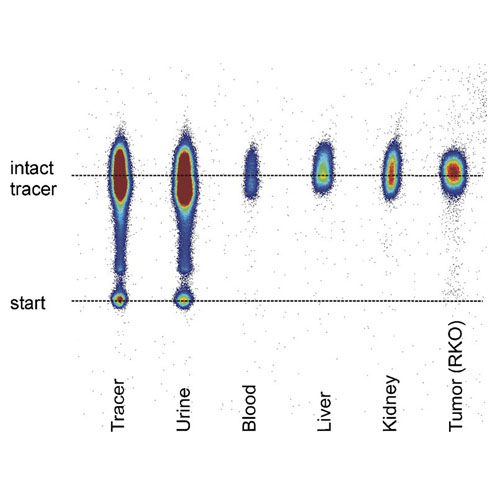
In vivo biokinetic and metabolic characterization of the 68Ga-labelled α5β1-selective peptidomimetic FR366
24-Oct-2015
European Journal of Nuclear Medicine and Molecular Imaging, Volume 43, Issue 5, pp 953-963, DOI 10.1007/s00259-015-3218-z
European Journal of Nuclear Medicine and Molecular Imaging, online article
Purpose Integrins are transmembrane receptors responsible for cell–cell adhesion and cell–extracellular matrix binding and play an important role in angiogenesis and tumour metastasis. For this reason, integrins are increasingly used as targets for molecular imaging. Up to now interest has mostly been focused on the integrin subtype αvβ3. However, targeting of other subtypes such as the integrin α5β1 is also of high interest due to its central role in colonization of metastatic cells, resistance of tumour cells to chemotherapy and ionizing radiation, and tumour aggressiveness. Recently, a highly active antagonist ligand (2,2′-(7-(1-
carboxy-4-(( 6 - ( ( 3 - ( 4 - ( ( ( S ) - 1-carboxy- 2 - (2- ( 3- guanidinobenzamido)acetamido)ethyl)carbamoyl)-3,5- dimethylphenoxy)propyl)amino)-6-oxohexyl)amino)-4- oxobutyl)-1,4,7-triazonane-1,4-diyl)diacetic acid, FR366).