Microsecond Timescale Protein Dynamics: a Combined Solid‐State NMR Approach
17-Nov-2017
ChemPhysChem, 19, 34 – 39, https://doi.org/10.1002/cphc.201701238
ChemPhysChem, online article
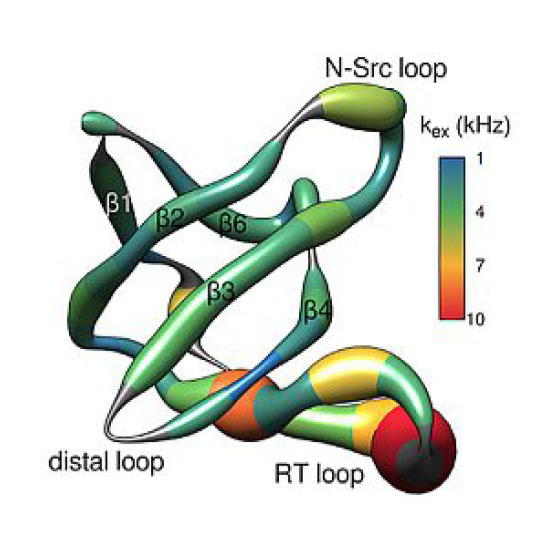
Conformational exchange in proteins is a major determinant in protein functionality. In particular, the μs–ms timescale is associated with enzymatic activity and interactions between biological molecules. We show here that a comprehensive data set of R1ρ relaxation dispersion profiles employing multiple effective fields and tilt angles can be easily obtained in perdeuterated, partly back‐exchanged proteins at fast magic‐angle spinning and further complemented with chemical‐exchange saturation transfer NMR experiments. The approach exploits complementary sources of information and enables the extraction of multiple exchange parameters for μs–ms timescale conformational exchange, most notably including the sign of the chemical shift differences between the ground and excited states.