Molecular basis for asymmetry sensing of siRNAs by the Drosophila Loqs-PD/Dcr-2 complex in RNA interference
13-Oct-2017
Nucleic Acids Research, Volume 45, Issue 21, Pages 12536–12550, https://doi.org/10.1093/nar/gkx886
Nucleic Acids Research, online article
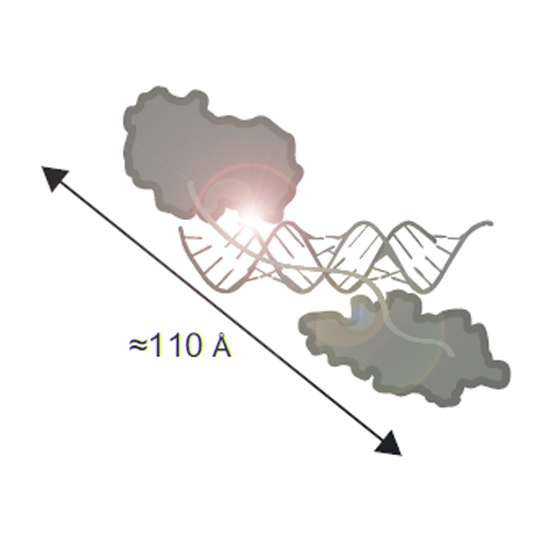
RNA interference defends against RNA viruses and retro-elements within an organism's genome. It is triggered by duplex siRNAs, of which one strand is selected to confer sequence-specificity to the RNA induced silencing complex (RISC). In Drosophila, Dicer-2 (Dcr-2) and the double-stranded RNA binding domain (dsRBD) protein R2D2 form the RISC loading complex (RLC) and select one strand of exogenous siRNAs according to the relative thermodynamic stability of base-pairing at either end. Through genome editing we demonstrate that Loqs-PD, the Drosophila homolog of human TAR RNA binding protein (TRBP) and a paralog of R2D2, forms an alternative RLC with Dcr-2 that is required for strand choice of endogenous siRNAs in S2 cells. Two canonical dsRBDs in Loqs-PD bind to siRNAs with enhanced affinity compared to miRNA/miRNA* duplexes. Structural analysis, NMR and biophysical experiments indicate that the Loqs-PD dsRBDs can slide along the RNA duplex to the ends of the siRNA. A moderate but notable binding preference for the thermodynamically more stable siRNA end by Loqs-PD alone is greatly amplified in complex with Dcr-2 to initiate strand discrimination by asymmetry sensing in the RLC.