On the use of the antibiotic chloramphenicol to target polypeptide chain mimics to the ribosomal exit tunnel
06-Sep-2013
Biochimie, 2013, http://dx.doi.org/10.1016/j.biochi.2013.06.004, Volume 95, Issue 9, Pages 1765–1772 published on 06.09.2013
Biochimie, online article
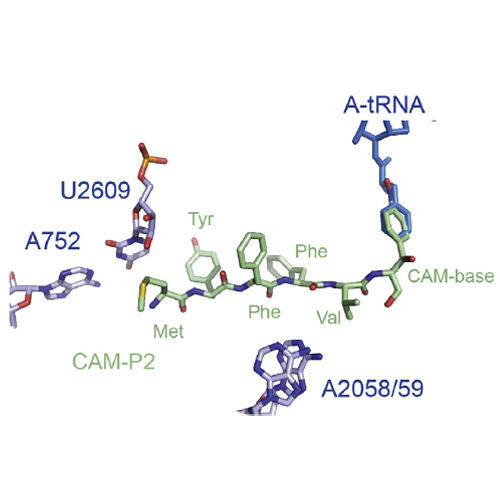
The ribosomal exit tunnel had recently become the centre of many functional and structural studies. Accumulated evidence indicates that the tunnel is not simply a passive conduit for the nascent chain, but a rather functionally important compartment where nascent peptide sequences can interact with the ribosome to signal translation to slow down or even stop. To explore further this interaction, we have synthesized short peptides attached to the amino group of a chloramphenicol (CAM) base, such that when bound to the ribosome these compounds mimic a nascent peptidyl-tRNA chain bound to the A-site of the peptidyltransferase center (PTC). Here we show that these CAM-peptides interact with the PTC of the ribosome while their effectiveness can be modulated by the sequence of the peptide, suggesting a direct interaction of the peptide with the ribosomal tunnel. Indeed, chemical footprinting in the presence of CAM-P2, one of the tested CAM-peptides, reveals protection of 23S rRNA nucleotides located deep within the tunnel, indicating a potential interaction with specific components of the ribosomal tunnel. Collectively, our findings suggest that the CAM-based peptide derivatives will be useful tools for targeting polypeptide chain mimics to the ribosomal tunnel, allowing their conformation and interaction with the ribosomal tunnel to be explored using further biochemical and structural methods.