pH-Dependent Dimerization and Salt-Dependent Stabilization of the N-terminal Domain of Spider Dragline Silk-Implications for Fiber Formation
03-Jan-2011
Angewandte Chemie, 2011, DOI: 10.1002/anie.201003795, Volume 50, Issue 1, pages 310–313 published on 03.01.2011
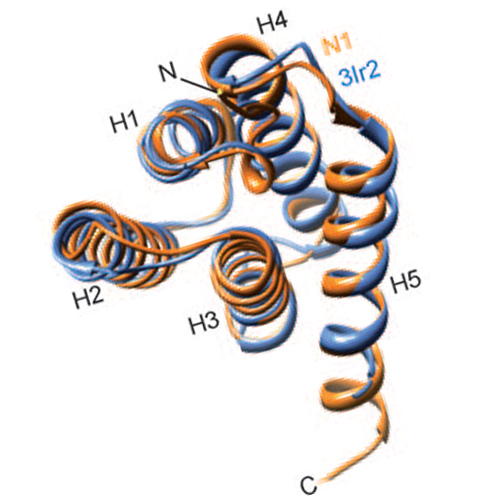
The formation of spider dragline silk is controlled by the relatively small C- and N-terminal domains of the spidroins. The formidable and unrivaled mechanical tensile strength of spider silk fibers is a result of the carefully matched assembly of polyalanine (polyA) or poly(glycinealanine) (polyGA) repeat sequences separated by GGX or GPGXX repeats, which are thought to confer elasticity to the thread.The correct alignment of polyA/polyGA sequences to form microcrystalline structures is controlled by the pH value, salt concentration, and shear-force-induced partial unfolding of the disulfide-bridged dimeric C-terminal domain.The N-terminal domain was also shown to be important for the pH-dependent assembly of fiber.Here, we use NMR spectroscopy and light-scattering techniques to show that the N-terminal domain of the major ampullate spider silk from Latrodectus hesperus (black widow spider) is mainly monomeric at neutral pH, as found in the spinning gland. The slight tendency to dimerize disappears under high salt conditions, as found in the gland. However, the Nterminal domain will dimerize at the lower pH value found in the spinning duct. Hence, acidification mainly controls the assembly of the N terminus, which is important for the formation of silk fiber, while high ionic strength stabilizes the monomeric N-terminal structure. The crystal structure of the N-terminal domain shows a homodimer with an antiparallel orientation of the subunit. In addition to this picture, our NMR data provide further evidence for the regulation and functional role of this domain in forming elongated silk threads.