Structural and biochemical characterization of the cell fate determining nucleotidyltransferase fold protein MAB21L1
08-Jun-2016
Scientific Reports, volume 6, Article number: 27498 (2016), https://doi.org/10.1038/srep27498
Scientific Reports, online article
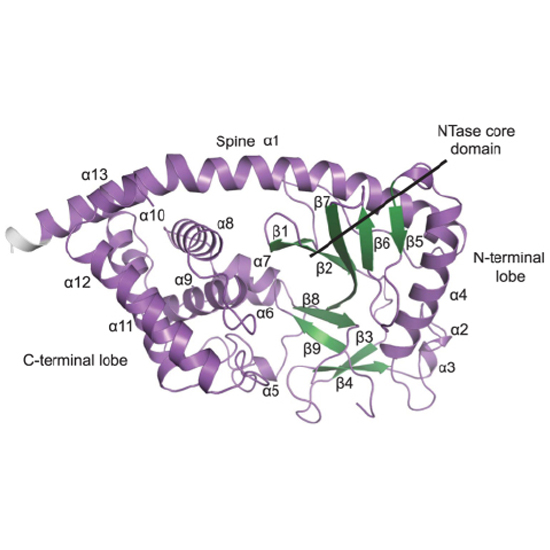
The exceptionally conserved metazoan MAB21 proteins are implicated in cell fate decisions and share considerable sequence homology with the cyclic GMP-AMP synthase. cGAS is the major innate immune sensor for cytosolic DNA and produces the second messenger 2′-5′, 3′-5′ cyclic GMP-AMP. Little is known about the structure and biochemical function of other proteins of the cGAS-MAB21 subfamily, such as MAB21L1, MAB21L2 and MAB21L3. We have determined the crystal structure of human full-length MAB21L1. Our analysis reveals high structural conservation between MAB21L1 and cGAS but also uncovers important differences. Although monomeric in solution, MAB21L1 forms a highly symmetric double-pentameric oligomer in the crystal, raising the possibility that oligomerization could be a feature of MAB21L1. In the crystal, MAB21L1 is in an inactive conformation requiring a conformational change - similar to cGAS - to develop any nucleotidyltransferase activity. Co-crystallization with NTP identified a putative ligand binding site of MAB21 proteins that corresponds to the DNA binding site of cGAS. Finally, we offer a structure-based explanation for the effects of MAB21L2 mutations in patients with eye malformations. The underlying residues participate in fold-stabilizing interaction networks and mutations destabilize the protein. In summary, we provide a first structural framework for MAB21 proteins.