Structural mechanism of ATP-dependent DNA binding and DNA end bridging by eukaryotic Rad50
19-Feb-2016
The EMBO Journal, Volume 35, Issue 7, Pages 759–772, DOI: 10.15252/embj.201592934
The EMBO Journal, online article
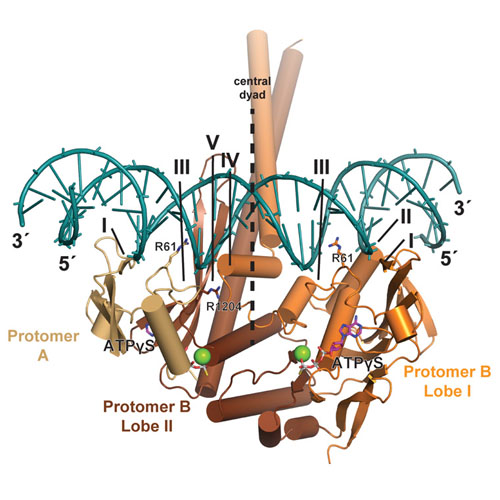
The Mre11–Rad50–Nbs1 (MRN) complex is a central factor in the repair of DNA double-strand breaks (DSBs). The ATP-dependent mechanisms of how MRN detects and endonucleolytically processes DNA ends for the repair by microhomology-mediated end-joining or further resection in homologous recombination are still unclear. Here, we report the crystal structures of the ATPγS-bound dimer of the Rad50NBD (nucleotide-binding domain) from the thermophilic eukaryote Chaetomium thermophilum (Ct) in complex with either DNA or CtMre11RBD (Rad50-binding domain) along with small-angle X-ray scattering and cross-linking studies. The structure and DNA binding motifs were validated by DNA binding experiments in vitro and mutational analyses in Saccharomyces cerevisiae in vivo. Our analyses provide a structural framework for the architecture of the eukaryotic Mre11–Rad50 complex. They show that a Rad50 dimer binds approximately 18 base pairs of DNA along the dimer interface in an ATP-dependent fashion or bridges two DNA ends with a preference for 3′ overhangs. Finally, our results may provide a general framework for the interaction of ABC ATPase domains of the Rad50/SMC/RecN protein family with DNA.