Structure of human mitochondrial RNA polymerase
25-Sep-2011
Nature Letter, 2011, 478, 269 - 73 published on 25.09.2011
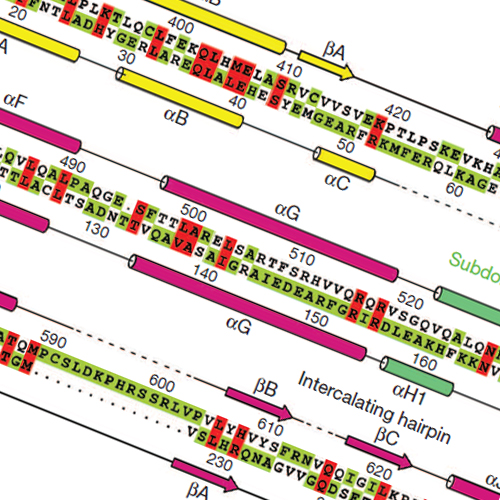
Transcription of the mitochondrial genome is performed by a single-subunit RNA polymerase (mtRNAP) that is distantly related to the RNAP of bacteriophage T7, the pol I family of DNA polymerases, and single-subunit RNAPs from chloroplasts1, 2, 3, 4. Whereas T7 RNAP can initiate transcription by itself, mtRNAP requires the factors TFAM and TFB2M for binding and melting promoter DNA5, 6, 7. TFAM is an abundant protein that binds and bends promoter DNA 15–40 base pairs upstream of the transcription start site, and stimulates the recruitment of mtRNAP and TFB2M to the promoter8, 9. TFB2M assists mtRNAP in promoter melting and reaches the active site of mtRNAP to interact with the first base pair of the RNA–DNA hybrid10. Here we report the X-ray structure of human mtRNAP at 2.5 Å resolution, which reveals a T7-like catalytic carboxy-terminal domain, an amino-terminal domain that remotely resembles the T7 promoter-binding domain, a novel pentatricopeptide repeat domain, and a flexible N-terminal extension. The pentatricopeptide repeat domain sequesters an AT-rich recognition loop, which binds promoter DNA in T7 RNAP, probably explaining the need for TFAM during promoter binding. Consistent with this, substitution of a conserved arginine residue in the AT-rich recognition loop, or release of this loop by deletion of the N-terminal part of mtRNAP, had no effect on transcription. The fingers domain and the intercalating hairpin, which melts DNA in phage RNAPs, are repositioned, explaining the need for TFB2M during promoter melting. Our results provide a new venue for the mechanistic analysis of mitochondrial transcription. They also indicate how an early phage-like mtRNAP lost functions in promoter binding and melting, which were provided by initiation factors in trans during evolution, to enable mitochondrial gene regulation and the adaptation of mitochondrial function to changes in the environment.