Systematic Backbone Conformational Constraints on a Cyclic Melanotropin Ligand Leads to Highly Selective Ligands for Multiple Melanocortin Receptors
28-Jul-2015
J. Med. Chem., 2015, 58 (16), pp 6359–6367, DOI: 10.1021/acs.jmedchem.5b00102
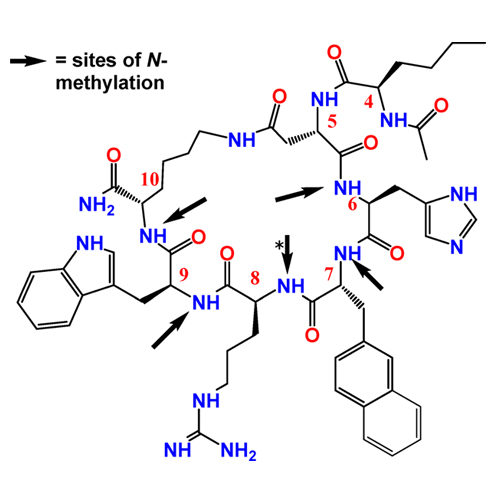
Human melanocortin receptors (hMCRs) have been challenging targets to develop ligands that are explicitly selective for each of their subtypes. To modulate the conformational preferences of the melanocortin ligands and improve the biofunctional agonist/antagonist activities and selectivities, we have applied a backbone N-methylation approach on Ac-Nle-c[Asp-His-d-Nal(2′)-Arg-Trp-Lys]-NH2 (Ac-Nle4-c[Asp5,d-Nal(2′)7,Lys10]-NH2), a nonselective cyclic peptide antagonist at hMC3R and hMC4R and an agonist at hMC1R and hMC5R. Systematic N-methylated derivatives of Ac-Nle4-c[Asp5,d-Nal(2′)7,Lys10]-NH2, with all possible backbone N-methylation combinations, have been synthesized and examined for their binding and functional activities toward melanocortin receptor subtypes 1, 3, 4, and 5 (hMCRs). Several N-methylated analogues are selective and potent agonists or antagonists for hMC1R or hMC5R or have selective antagonist activity for hMC3R. The selective hMC1R ligands show strong binding for human melanoma cells. We have also discovered the first universal antagonist (compound 19) for all subtypes of hMCRs.