The archaeal RNA polymerase subunit P and the eukaryotic polymerase subunit Rpb12 are interchangeable in vivo and in vitro
18-Dec-2008
Mol. Microbiol., 2009, 71(4), 989-1002, doi:10.1111/j.1365-2958.2008.06577.x published on 18.12.2008
Molecular Microbiology, online article
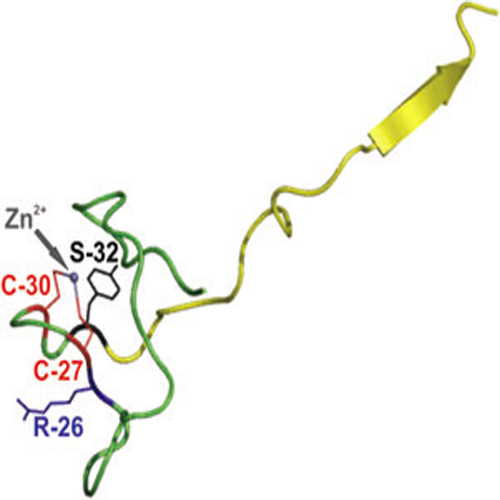
The general subunit of all three eukaryotic RNA polymerases, Rpb12, and subunit P of the archaeal enzyme show sequence similarities in their N-terminal zinc ribbon and some highly conserved residues in the C-terminus. We report here that archaeal subunit P under the control of a strong yeast promoter could complement the lethal phenotype of a RPB12 deletion mutant and that subunit Rpb12 from yeast can functionally replace subunit P during reconstitution of the archaeal RNA polymerase. The ΔP enzyme is unable to form stable open complexes, but can efficiently extend a dinucleotide on a premelted template or RNA on an elongation scaffold. This suggests that subunit P is directly or indirectly involved in promoter opening. The activity of the ΔP enzyme can be rescued by the addition of Rpb12 or subunit P to transcription reactions. Mutation of cysteine residues in the zinc ribbon impair the activity of the enzyme in several assays and this mutated form of P is rapidly replaced by wild-type P in transcription reactions. The conserved zinc ribbon in the N-terminus seems to be important for proper interaction of the complete subunit with other RNA polymerase subunits and a 17-amino-acid C-terminal peptide is sufficient to support all basic RNA polymerase functions in vitro.