The Role of 23S Ribosomal RNA Residue A2451 in Peptide Bond Synthesis Revealed by Atomic Mutagenesis
24-Apr-2008
Chemistry & Biology, 2008, Vol. 15, DOI: 10.1016/j.chembiol.2008.03.014 published on 24.04.2008
Chemistry & Biology; online article
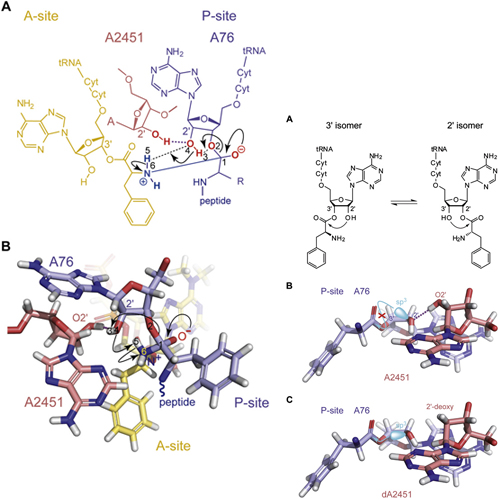
Peptide bond formation is a fundamental reaction in biology, catalyzed by the ribosomal peptidyl-transferase ribozyme. Although all active-site 23S ribosomal RNA nucleotides are universally conserved, atomic mutagenesis suggests that these nucleobases do not carry functional groups directly involved in peptide bond formation. Instead, a single ribose 20-hydroxyl group at A2451 was identified to be of pivotal importance. Here, we altered the chemical characteristics by replacing its 20-hydroxyl with selected functional groups and demonstrate that hydrogen donor capability is essential for transpeptidation. We propose that the A2451-20-hydroxyl directly hydrogen bonds to the P-site tRNA-A76 ribose. This promotes an effective A76 ribose C20-endo conformation to support amide synthesis via a proton shuttle mechanism. Simultaneously, the direct interaction of A2451 with A76 renders the intramolecular transesterification of the peptide from the 30- to 20-oxygen unfeasible, thus promoting effective peptide bond synthesis.