Variation of Specific Activities of Ga-68-Aquibeprin and Ga-68-Avebetrin Enables Selective PET-Imaging of Different Expression Levels of Integrins α5β1 and αvβ3
05-May-2016
THE JOURNAL OF NUCLEAR MEDICINE, Vol. 57, No. 10, 1618–1624, doi: 10.2967/jnumed.116.173948
J Nucl Med., online article
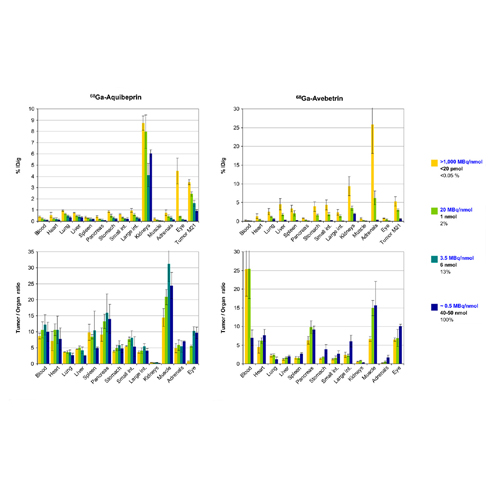
Ga-68-Aquibeprin and Ga-68-Avebetrin are tracers for selective in-vivo mapping of integrins α5β1 and αvβ3, respectively, by positron emission tomography (PET). As both tracers exhibit very high affinity to their respective targets, the aim of this study was to investigate the influence of the specific activity of preparations of both tracers on in vivo imaging results. Methods: Fully automated Ga-68 labelling of 0.3 nmol of Aquibeprin or Avebetrin was done using buffered eluate fractions (600–800 MBq, pH 2) of a SnO2-based generator, affording the radiopharmaceuticals with specific activities >1,000 MBq/nmol. Lower values ranging from 150 to 0.4 MBq/nmol were adjusted by addition of inactive compound (approx. 0.15 to 50 nmol) to the injected activity (~ 20 MBq for PET, 5–7 MBq for biodistribution). For in vivo experiments, 6–12 weeks old, female SCID mice bearing M21 xenografts (human melanoma, expressing both integrins α5β1 and αvβ3) were used. The expression density of integrin β3 was determined by immunohistochemistry on paraffine slices. Results: For mass doses (specific activities) of < 20 pmol (>1,000 MBq/nmol) and 1 nmol (20 MBq/nmol) per mouse, respectively, uptakes of Ga-68-Aquibeprin / Ga-68-Avebetrin in M21 tumors dropped from 5.3 / 3.5 % ID/g to 3.0 / 2.4 % ID/g, respectively. Applying < 20 pmol, high uptakes of Ga-68-Aquibeprin in eyes (4.5 % ID/g) or Ga-68-Avebetrin in adrenals (25.9 % ID/g), respectively, were found, which were reduced by 90 and 65% (0.44 and 6.2 % ID/g, respectively), for doses of 1 nmol. Highest tumor-to-tissue ratios were observed both in ex-vivo biodistribution and PET for comparably large doses, e.g., 6 nmol (0.65 mg/kg) Ga-68-Aquibeprin per mouse (3.5 MBq/nmol). Conclusion: Presumably owing to their high affinities, Ga-68-Aquibeprin and Ga-68-Avebetrin allow for selective addressing of target sites with different integrin expression levels by virtue of adjusting specific activity, which can be exploited for visualization of low-level target expression or optimization of tumor-to-background contrast.