A Total Synthesis Prompts the Structure Revision of Haouamine B
30-Apr-2012
J. Am. Chem. Soc., 2012, DOI: 10.1021/ja301326k, 134 (22), pp 9291–9295 published on 30.04.2012
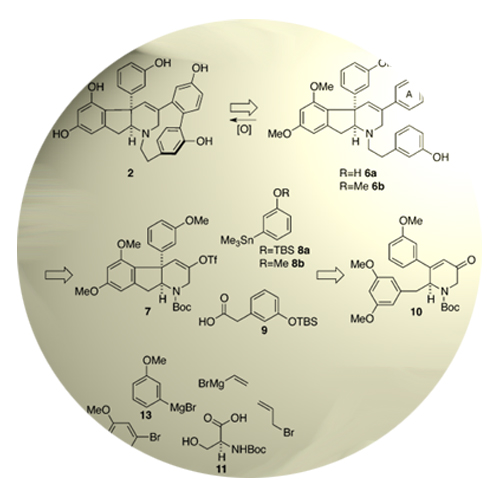
A concise asymmetric approach to the indeno-tetrahydropyridine core of the unusual alkaloid haouamine B allowed for an investigation of a biomimetic oxidative phenol coupling as a proposed biosynthetic step, and ultimately provided access to the published structure of the natural product. As a consequence of our synthetic studies, the structure of haouamine B has been revised.