Characterization of a novel cytochrome cGJ as the electron acceptor of XoxF-MDH in the thermoacidophilic methanotroph Methylacidiphilum fumariolicum SolV
04-Apr-2019
Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics, Volume 1867, Issue 6, Pages 595-603, https://doi.org/10.1016/j.bbapap.2019.04.001
BBA, online article
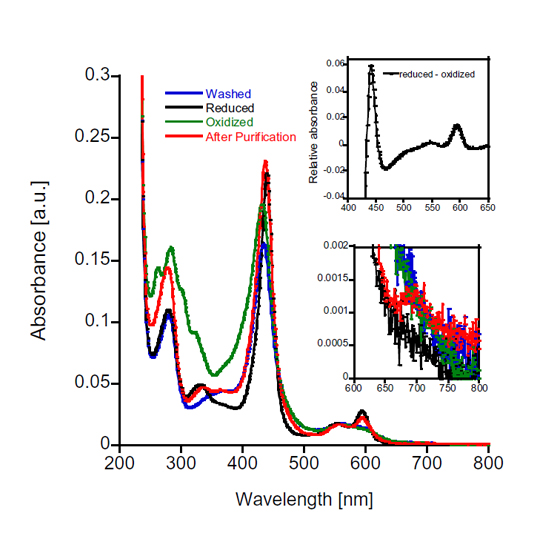
Methanotrophs play a prominent role in the global carbon cycle, by oxidizing the potent greenhouse gas methane to CO2. Methane is first converted into methanol by methane monooxygenase. This methanol is subsequently oxidized by either a calcium-dependent MxaF-type or a lanthanide-dependent XoxF-type methanol dehydrogenase (MDH). Electrons from methanol oxidation are shuttled to a cytochrome redox partner, termed cytochrome cL. Here, the cytochrome cL homolog from the thermoacidophilic methanotroph Methylacidiphilum fumariolicum SolV was characterized. SolV cytochrome cGJ is a fusion of a XoxG cytochrome and a periplasmic binding protein XoxJ. Here we show that XoxGJ functions as the direct electron acceptor of its corresponding XoxF-type MDH and can sustain methanol turnover, when a secondary cytochrome is present as final electron acceptor. SolV cytochrome cGJ (XoxGJ) further displays a unique, red-shifted absorbance spectrum, with a Soret and Q bands at 440, 553 and 595 nm in the reduced state, respectively. VTVH-MCD spectroscopy revealed the presence of a low spin iron heme and the data further shows that the heme group exhibits minimal ruffling. The midpoint potential Em,pH7 of +240 mV is similar to other cytochrome cL type proteins but remarkably, the midpoint potential of cytochrome cGJ was not influenced by lowering the pH. Cytochrome cGJ represents the first example of a cytochrome from a strictly lanthanide-dependent methylotrophic microorganism.