Chemical Proteomics Uncovers EPHA2 as a Mechanism of Acquired Resistance to Small Molecule EGFR Kinase Inhibition
12-May-2015
J. Proteome Res., 14 (6), pp 2617–2625, DOI: 10.1021/acs.jproteome.5b00161
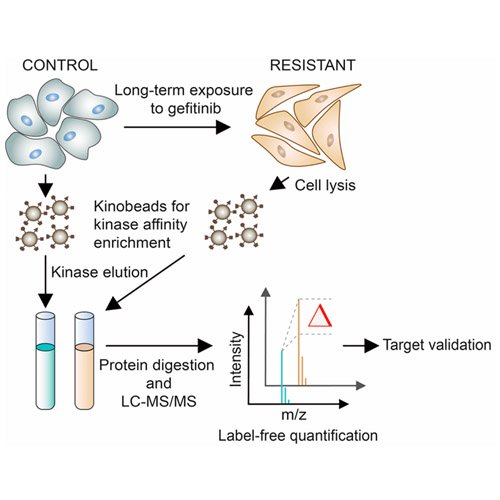
Tyrosine kinase inhibitors (TKIs) have become an important therapeutic option for treating several forms of cancer. Gefitinib, an inhibitor of the epidermal growth factor receptor (EGFR), is in clinical use for treating non-small cell lung cancer (NSCLC) harboring activating EGFR mutations. However, despite high initial response rates, many patients develop resistance to gefitinib. The molecular mechanisms of TKI resistance often remain unclear. Here, we describe a chemical proteomic approach comprising kinase affinity purification (kinobeads) and quantitative mass spectrometry for the identification of kinase inhibitor resistance mechanisms in cancer cells. We identified the previously described amplification of MET and found EPHA2 to be more than 10-fold overexpressed (p < 0.001) in gefitinib-resistant HCC827 cells suggesting a potential role in developing resistance. siRNA-mediated EPHA2 knock-down or treating cells with the multikinase inhibitor dasatinib restored sensitivity to gefitinib. Of all dasatinib targets, EPHA2 exhibited the most drastic effect (p < 0.001). In addition, EPHA2 knockdown or ephrin-A1 treatment of resistant cells decreased FAK phosphorylation and cell migration. These findings confirm EPHA2 as an actionable drug target, provide a rational basis for drug combination approaches, and indicate that chemical proteomics is broadly applicable for the discovery of kinase inhibitor resistance.