Homotypic Interaction and Amino Acid Distribution of Unilaterally Conserved Transmembrane Helices
13-Jul-2012
Journal of Molecular Biology, 2012, doi:10.1016/j.jmb.2012.04.008, Pages 251–257 published on 13.07.2012
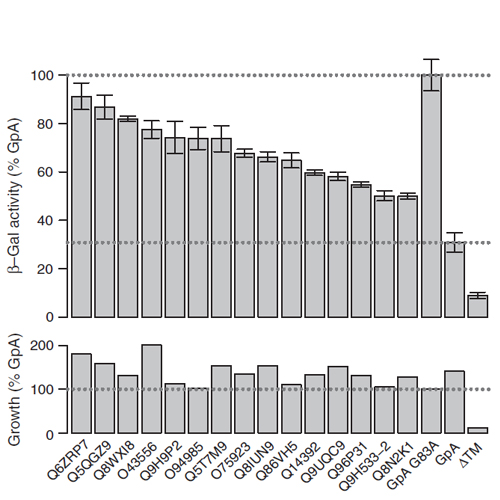
Formation of non-covalent functional complexes of integral membrane proteins is frequently supported by sequence-specific interaction of their transmembrane helices. Here, we aligned human single-span membrane proteins with orthologs from other eukaryotes. We find that almost half of the human single-span membrane proteins contain a transmembrane helix that exhibits significant non-random unilateral conservation. Furthermore, unilateral conservation of transmembrane domains (TMDs) correlates well with their ability to self-interact. Glycine, polar non-ionizable, and aromatic amino acids are overrepresented in conserved versus non-conserved helix faces. Hence, our genome-wide analysis indicates that these amino acid types generally support interaction of single-span membrane protein TMDs.