Phosphorothioation of Oligonucleotides Strongly Influences the Inhibition of Bacterial (M.HhaI) and Human(Dnmt1) DNA Methyltransferases
16-Feb-2009
ChemBioChem, 2009, 10, 728-734 published on 16.02.2009
ChemBioChem, online article
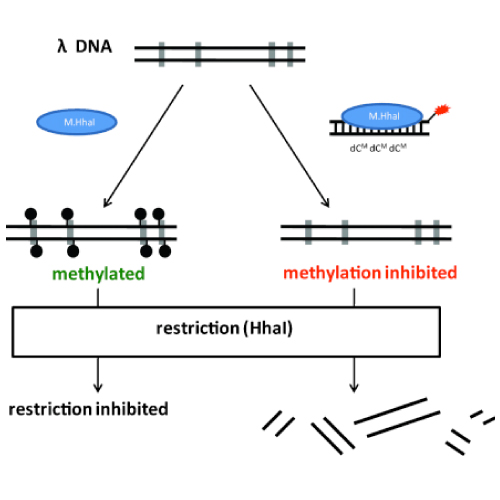
The cytidine analogue 5-fluoro-2-deoxycytidine (dCF) is a mechanism-based inhibitor of DNA methyltransferases. We report the synthesis of short 18-mer dsDNA oligomers containing a triple-hemimethylated CpG motive as a recognition sequence for the human methyltransferase Dnmt1. The DNA strands carry within these CpG islands dCF building blocks that function as mechanism-based inhibitors of the analyzed methyltransferases. In addition, we replaced the phosphodiester backbones at defined positions by phosphorothioates. These hypermodified DNA strands were investigated as inhibitors of the DNA methyltransferases M.HhaI and Dnmt1 in vitro. We could show that both methylases behave substantially differently in respect to the amount of DNA backbone modification.