Protein Iodination by Click Chemistry
31-Mar-2009
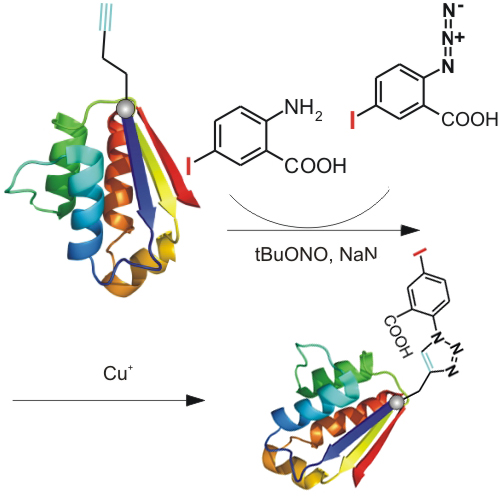
Upon development of radioimmunoassay techniques,[1] radioiodination of peptides and proteins has gradually become an indispensable tool for controlling and monitoring protein and peptide functions in vivo and in vitro, and for studying ligand–receptor interactions, ligand uptake and clearance. In addition, protein iodination has developed into a useful technique for heavy-metal modification in crystallographic analysis,[2] and serves as a valuable alternative to the expression of proteins with chalcogen amino acid analogues, such as selenomethionine, telluromethionine and selenocysteine.[3] Direct iodination of peptides and proteins at histidine, and more efficiently at tyrosine residues, under the various oxidative conditions is accompanied by modification of sensitive amino acid residues, such as methionine, cysteine and particularly tryptophan.[4] Indirect iodination methods that are based mainly on conjugation of peptides and folded proteins at surface exposed amine functions with iodinated N-hydroxysuccinimidyl-3-(4-hydroxyphenyl) propionate (Bolton–Hunter reagent[5]), and more recently with N-succinimidyl 3-iodobenzoate, have, therefore, been proposed.