Temperature and pressure dependence of protein stability: The engineered fluorescein-binding lipocalin FluA shows an elliptic phase diagram
15-Apr-2008
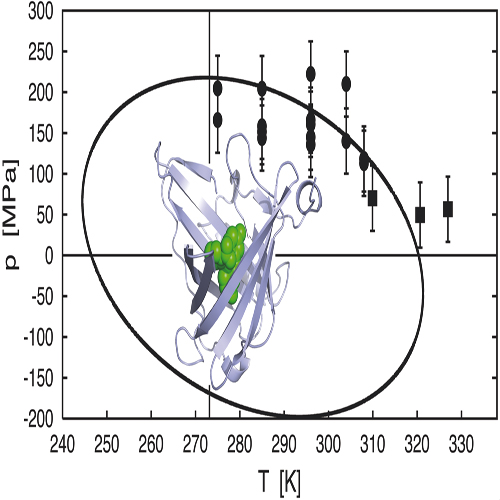
We have measured the equilibrium constant for the denaturation transition of the engineered fluorescein-binding lipocalin FluA as a function of pressure and temperature, taking advantage of the fact that the ligand’s fluorescence is almost fully quenched when complexed with the folded protein, but reversibly reappears on denaturation. From the equilibrium constant as a function of pressure and temperature all of the involved thermodynamic parameters of protein folding, in particular the changes in entropy and volume, compressibility, thermal expansion, and specific heat, were deduced in a global fitting procedure. Assuming that these parameters are independent of temperature and pressure, we can demonstrate from the ratio of Δβ, Δα², ΔCp that the phase diagram of protein folding assumes an elliptic shape. urthermore, we can show that the thermodynamic condition for such an elliptic phase diagram is related to the degree of correlation between the fluctuations of the changes in volume and enthalpy at the phase boundary. For the protein investigated this correlation is low, as generally expected for highly degenerate systems. Our study suggests that the elliptic phase diagram is a consequence of the inherent conformational disorder of proteins and that it may be viewed as the thermodynamic manifestation of the high degeneracy of conformational energies that is characteristic for this class of macromolecules.