The 26S proteasome: assembly and function of a destructive machine
10-Jun-2010
Trends in Biochemical Science, 2010, doi:10.1016/j.tibs.2010.05.005, Volume 35, Issue 11, Pages 634-642 published on 10.06.2010
Trends in Biochemical Science, online article
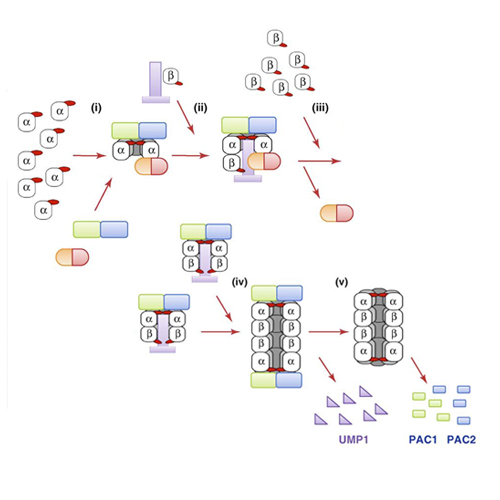
The heart of the ubiquitin-mediated degradation pathway, the 26S proteasome, endoproteolytically cleaves most intracellular proteins, thereby maintaining biological homeostasis and regulating many crucial processes in the cell. This hydrolyzing machine comprises more than 30 different subunits, which perform different functions including the recognition, unfolding, translocating and cleavage of protein substrates. Thus, careful assemblage and regulation of the 26S proteasome is essential to ensure correct positioning and function of each subunit, thereby preserving the delicate cellular balance between protein synthesis and degradation. Here, we review the most current research on the 26S proteasome assembly pathway, and describe the mechanism used by the cell to manage the complex structure and functions of the proteasome.