The Discovery of 5-Formylcytosine in Embryonic Stem Cell DNA
25-Jul-2011
Angewandte Chemie, 2011, DOI: 10.1002/ange.201103899, Volume 123, Issue 31, pages 7146–7150 published on 25.07.2011
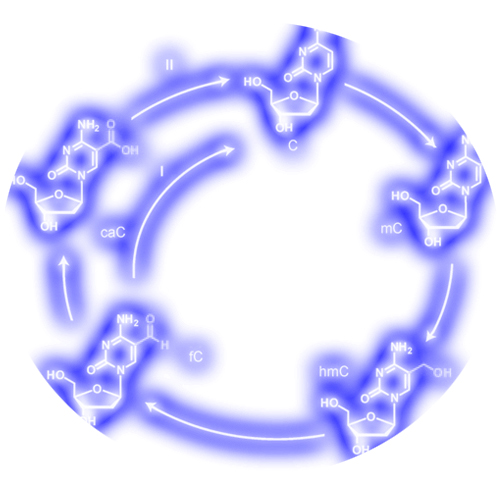
Cellular development requires the silencing and activation of specific gene sequences in a well-orchestrated fashion. Transcriptional gene silencing is associated with the clustered methylation of cytosine bases (C) in CpG units of promoters. The methylation occurs at position C5 of cytosine to give 5- methylcytosine (mC) with the help of special DNA methyltransferases (DNMT). The DNA methylome is significantly reprogrammed at various stages during early development, during the development of primordial germ cells, or later in a locus-specific way at postdevelopmental stages. Decreasing levels of mC can be established passively by successive rounds of DNA replication in the absence of methyltransferases. Active demethylation, in contrast, is proposed to be a process in which the mC bases are directly converted back into unmodified cytosines in the genome. The recent discovery that mC can be further oxidized to hydroxymethylcytosine (hmC) with the help of TET enzymes has led to the idea that hmC is connected to epigenetic reprogramming, maybe as an intermediate in an, as yet controversial, active demethylation process. Indeed recent data suggest that active demethylation in postdevelopmental phases may proceed through deamination of hmC to give 5-hydroxymethyluridine (hmU), which is then removed from the genome with the help of the base excision repair (BER) system. Chemically, an attractive alternative mechanism for a more global active demethylation could be envisioned through further oxidation of hmC to give either 5- formylcytosine (fC) or 5-carboxylcytosine (caC) followed by elimination of a formyl or carboxyl group, respectively . Although such an oxidative active demethylation pathway with hmC as the starting point has been frequently postulated, none of the further oxidized bases (fC, caC) have so far been detected.