The Inflammasome Drives GSDMD-Independent Secondary Pyroptosis and IL-1 Release in the Absence of Caspase-1 Protease Activity
26-Dec-2017
Cell Reports, Volume 21, Issue 13, p3846–3859, DOI: https://doi.org/10.1016/j.celrep.2017.12.018
Cell Reports, online article
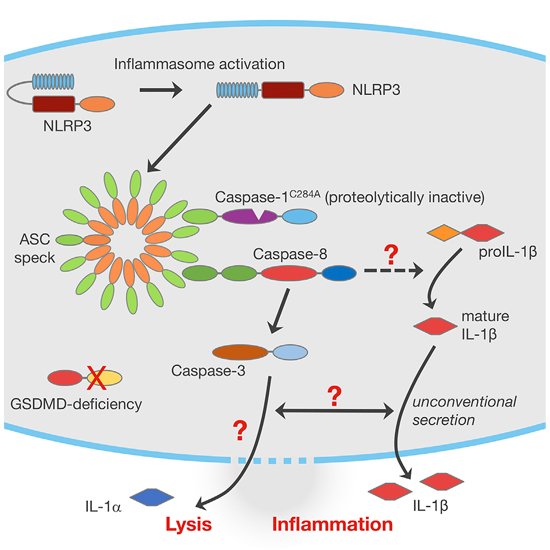
Inflammasomes activate the protease caspase-1, which cleaves interleukin-1β and interleukin-18 to generate the mature cytokines and controls their secretion and a form of inflammatory cell death called pyroptosis. By generating mice expressing enzymatically inactive caspase-1C284A, we provide genetic evidence that caspase-1 protease activity is required for canonical IL-1 secretion, pyroptosis, and inflammasome-mediated immunity. In caspase-1-deficient cells, caspase-8 can be activated at the inflammasome. Using mice either lacking the pyroptosis effector gasdermin D (GSDMD) or expressing caspase-1C284A, we found that GSDMD-dependent pyroptosis prevented caspase-8 activation at the inflammasome. In the absence of GSDMD-dependent pyroptosis, the inflammasome engaged a delayed, alternative form of lytic cell death that was accompanied by the release of large amounts of mature IL-1 and contributed to host protection. Features of this cell death modality distinguished it from apoptosis, suggesting it may represent a distinct form of pro-inflammatory regulated necrosis.