The Two Faces of Potent Antitumor Duocarmycin-Based Drugs: A Structural Dissection Reveals Disparate Motifs for DNA versus Aldehyde Dehydrogenase 1 Affinity
01-Jul-2013
Angewandte Chemie, 2013, DOI: 10.1002/anie.201208941, Volume 52, Issue 27, pages 6921–6925 published on 01.07.2013
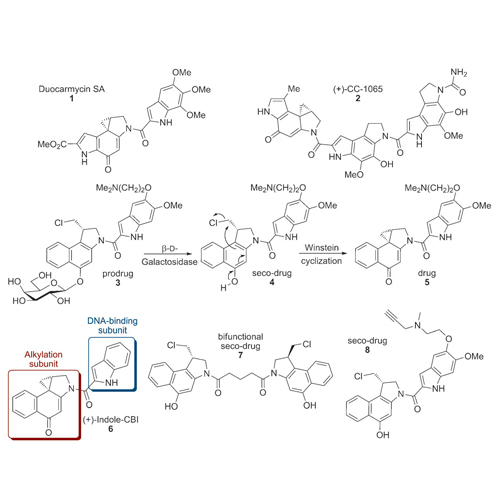
Duocarmycin-derived seco-cyclopropabenzindole (CBI) drugs have been shown to bind DNA and an aldehyde dehydrogenase (ALDH1A1) in lung cancer cells. The removal of the DNA-binding indole moiety results in a CBI compound that does not bind to DNA in whole cells but still exhibits remarkable cytotoxicity. This CBI compound has an increased affinity for ALDH1A1. Rh=rhodamine.