CipA and CipB as Scaffolds To Organize Proteins into Crystalline Inclusions
10-Feb-2017
ACS Synth. Biol., 6 (5), pp 826–836, DOI: 10.1021/acssynbio.6b00323
ACS Synth. Biol., online article
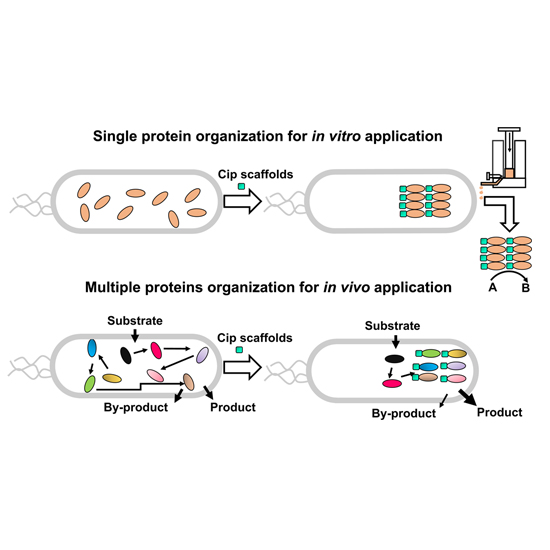
Natural and synthetic scaffolds support enzyme organization in complexes, and they regulate their function and activity. Here we report that CipA and CipB, two small proteins that form protein crystalline inclusions (PCIs) in the cytoplasm of Photorhabdus luminescens, can be utilized as scaffolds to efficiently incorporate exogenous proteins into PCIs. We demonstrate that Cip-tagged GFP is assembled into fluorescent PCIs in P. luminescens and that in Escherichia coli Cip scaffolds can organize GFP or/and LacZ into bioactive PCIs, which could easily be isolated for in vitro catalysis. To explore its in vivo application further, we used CipA to bring together multiple enzymes (Vio enzymes) of the violacein biosynthetic pathway. The resulting complexes were found to produce significantly higher yields of violacein and fewer byproducts than did Vio enzymes in solution. Hence, Cip scaffolds should be widely applicable to biotechnological processes both in vitro and in vivo.