DNA-binding directs the localization of a membrane-integrated receptor of the ToxR family
04-Jan-2019
COMMUNICATIONS BIOLOGY, (2019) 2:4, https://doi.org/10.1038/s42003-018-0248-7
COMMUNICATIONS BIOLOGY, online article
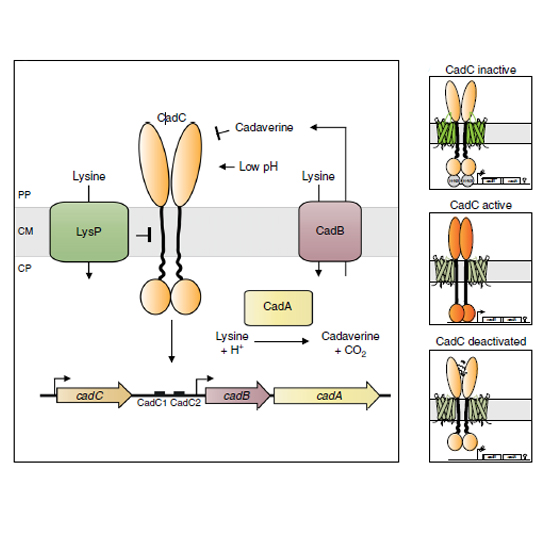
All living cells have a large number of proteins that are anchored with one transmembrane helix in the cytoplasmic membrane. Almost nothing is known about their spatiotemporal organization in whole cells. Here we report on the localization and dynamics of one representative, the pH sensor and transcriptional regulator CadC in Escherichia coli. Fluorophore-tagged CadC was detectable as distinct cluster only when the receptor was activated by external stress, which results in DNA-binding. Clusters immediately disappeared under non-stress conditions. CadC variants that mimic the active state of CadC independent of environmental stimuli corroborated the correlation between CadC clustering and binding to the DNA, as did altering the number or location of the DNA-binding site(s) in whole cells. These studies reveal a novel diffusion-and-capture mechanism to organize a membrane-integrated receptor dependent on the DNA in a rod-shaped bacterium.