Insights into the DNA-binding mechanism of a LytTR-type transcription regulator
24-Mar-2016
Bioscience Reports, 36, e00326, DOI: 10.1042/BSR20160069
Bioscience Reports, online article
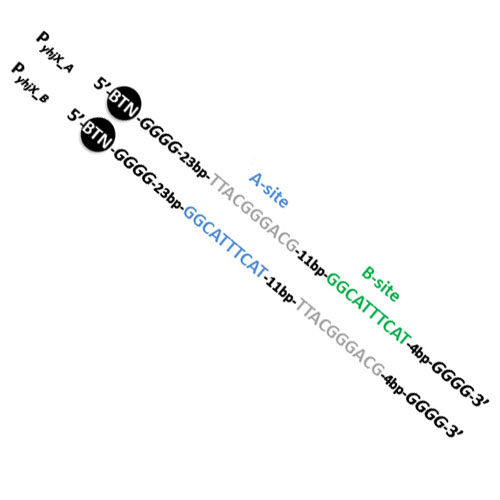
Most bacterial response regulators make contact with DNA through a recognition α-helix in their DNA-binding domains. An emerging class of response regulators interacts with DNA via a relatively novel type of binding domain, called the LytTR domain, which is mainly composed of beta-strands. YpdB belongs to this latter class, is part of a nutrient-sensing network in Escherichia coli and triggers expression of its only target gene, yhjX, in response to extracellular pyruvate. Expression of yhjX mainly occurs in the late exponential growth phase, and in a pulsed manner. Although the DNA-binding sites for YpdB are well defined, exactly how YpdB initiates pulsed gene expression has remained elusive. To address this question, we measured the binding kinetics of wild-type YpdB and the phosphomimetic mutant YpdB-D53E to the yhjX promoter region (PyhjX) using surface plasmon resonance spectroscopy combined with interaction map® analysis. Both YpdB and YpdB-D53E bound as monomers to the tandem-repeat sequences in the promoter, with YpdB-D53E displaying a higher maximal binding rate than YpdB. Furthermore, we identified a high-affinity (A-site) and a low-affinity binding site (B-site) within the yhjX promoter. Only YpdB-D53E utilizes an "AB-BA" DNA-binding mechanism, involving sequential and cooperative promoter binding, and rapid, successive promoter clearance. We propose that response-regulator phosphorylation, in combination with the cycle of cooperative DNA-binding and rapid promoter clearance just described, can account for pulsed gene expression.