Interaction Analysis of a Two-Component System Using Nanodiscs
16-Feb-2016
Plos One, 11 (2), DOI:10.1371/journal.pone.0149187
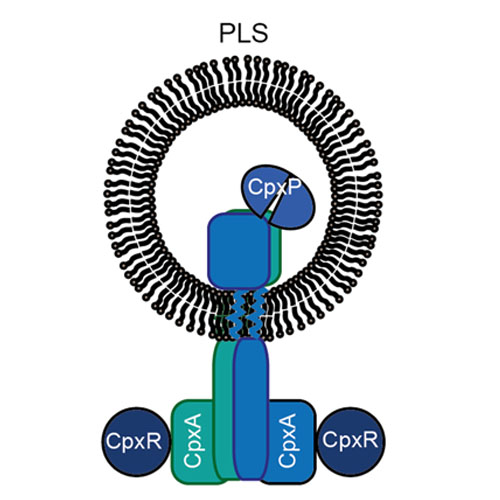
Two-component systems are the major means by which bacteria couple adaptation to environmental changes. All utilize a phosphorylation cascade from a histidine kinase to a response regulator, and some also employ an accessory protein. The system-wide signaling fidelity of two-component systems is based on preferential binding between the signaling proteins. However, information on the interaction kinetics between membrane embedded histidine kinase and its partner proteins is lacking. Here, we report the first analysis of the interactions between the full-length membrane-bound histidine kinase CpxA, which was reconstituted in nanodiscs, and its cognate response regulator CpxR and accessory protein CpxP. Using surface plasmon resonance spectroscopy in combination with interaction map analysis, the affinity of membrane-embedded CpxA for CpxR was quantified, and found to increase by tenfold in the presence of ATP, suggesting that a considerable portion of phosphorylated CpxR might be stably associated with CpxA in vivo. Using microscale thermophoresis, the affinity between CpxA in nanodiscs and CpxP was determined to be substantially lower than that between CpxA and CpxR. Taken together, the quantitative interaction data extend our understanding of the signal transduction mechanism used by two-component systems.