Life span extension by targeting a link between metabolism and histone acetylation in Drosophila
18-Jan-2016
EMBO reports, DOI: 10.15252/embr.201541132
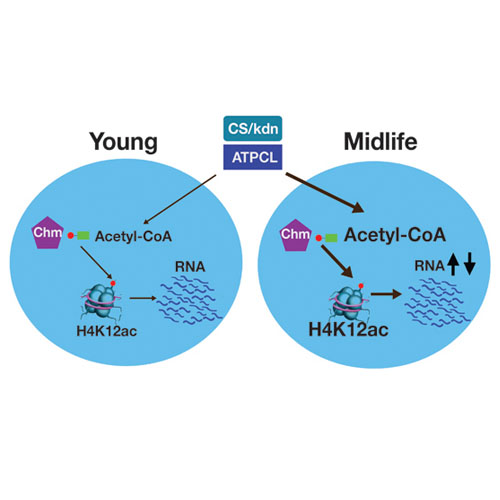
Old age is associated with a progressive decline of mitochondrial function and changes in nuclear chromatin. However, little is known about how metabolic activity and epigenetic modifications change as organisms reach their midlife. Here, we assessed how cellular metabolism and protein acetylation change during early aging in Drosophila melanogaster. Contrary to common assumptions, we find that flies increase oxygen consumption and become less sensitive to histone deacetylase inhibitors as they reach midlife. Further, midlife flies show changes in the metabolome, elevated acetyl-CoA levels, alterations in protein—notably histone—acetylation, as well as associated transcriptome changes. Based on these observations, we decreased the activity of the acetyl-CoA-synthesizing enzyme ATP citrate lyase (ATPCL) or the levels of the histone H4 K12-specific acetyltransferase Chameau. We find that these targeted interventions both alleviate the observed aging-associated changes and promote longevity. Our findings reveal a pathway that couples changes of intermediate metabolism during aging with the chromatin-mediated regulation of transcription and changes in the activity of associated enzymes that modulate organismal life span.