Aromatic Foldamer Helices as α‐Helix Extended Surface Mimetics
10-Sep-2020
Chem. Eur. J. 2020, 26, 17366 – 17370, doi.org/10.1002/chem.202004064
Chem. Eur. J., online article
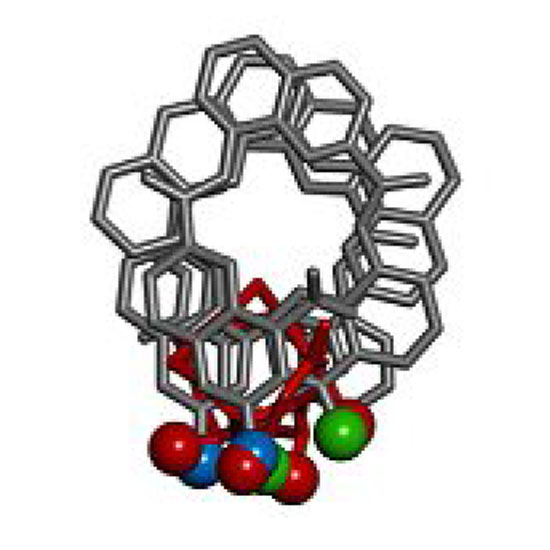
Helically folded aromatic oligoamide foldamers have a size and geometrical parameters very distinct from those of α‐helices and are not obvious candidates for α‐helix mimicry. Nevertheless, they offer multiple sites for attaching side chains. It was found that some arrays of side chains at the surface of an aromatic helix make it possible to mimic extended α‐helical surfaces. Synthetic methods were developed to produce quinoline monomers suitably functionalized for solid phase synthesis. A dodecamer was prepared. Its crystal structure validated the initial design and showed helix bundling involving the α‐helix‐like interface. These results open up new uses of aromatic helices to recognize protein surfaces and to program helix bundling in water.