- Home ·
- Publications ·
- Research Area F ·
- 2008 ·
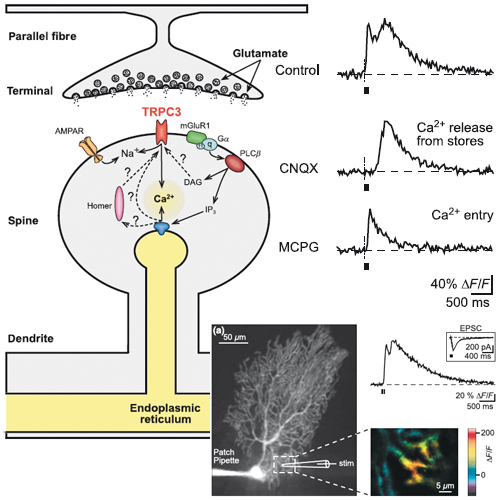
Mechanisms of metabotropic glutamate receptor-mediated synaptic signalling in cerebellar Purkinje cells
28-Oct-2008
J. Hartmann, A. Konnerth
The metabotropic glutamate receptors type 1 (mGluR1s) are required for a normal function of the mammalian cerebellum. These G-protein-coupled receptors are abundantly expressed in the principle cerebellar cells, namely the Purkinje neurones. Under physiological conditions, mGluR1s are activated during repetitive activity of both afferent glutamatergic synaptic inputs provided by the climbing and parallel fibres respectively. Unlike the common ionotropic glutamate receptors that underlie rapid synaptic excitation, mGluR1s produce a complex post-synaptic response consisting of a Ca2+-release signal from intracellular stores and a slow excitatory postsynaptic potential. While it is well established that the mGluR1-dependent Ca2+-release signal from intracellular stores involves the activation of inositol- trisphosphate receptors, the mechanisms underlying the slow synaptic excitation remained unclear. Here we will review recent evidence indicating an essential role of C-type transient receptor potential (TRPC) cation channels, especially that of the subunit TRPC3, for the generation of the mGluR1-dependent synaptic current. For the signalling pathways underlying both, Ca2+-release from intracellular stores and the slow synaptic potential, we present current knowledge about the activators, downstream effectors and possible roles for mGluR1-dependent signalling in Purkinje neurones.