A key role for cyclic nucleotide gated (CNG) channels in cGMP-related retinitis pigmentosa
05-Jan-2011
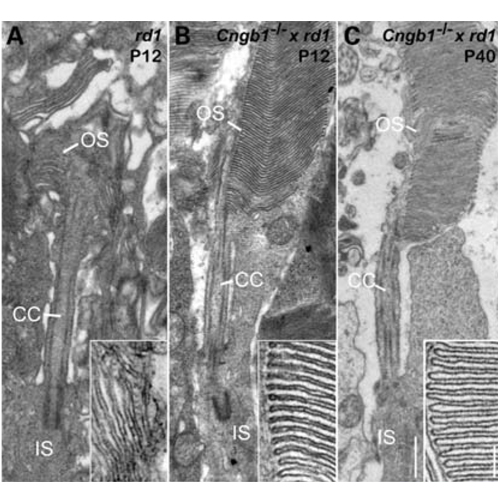
The rd1 natural mutant is one of the first and probably the most commonly studied mouse model for retinitis pigmentosa (RP), a severe and frequently blinding human retinal degeneration. In several decades of research, the link between the increase in photoreceptor cGMP levels and the extremely rapid cell death gave rise to a number of hypotheses. Here, we provide clear evidence that the presence of cyclic nucleotide gated (CNG) channels in the outer segment membrane is the key to rod photoreceptor loss. In Cngb1−/− × rd1 double mutants devoid of regular CNG channels, cGMP levels are still pathologically high, but rod photoreceptor viability and outer segment morphology are greatly improved. Importantly, cone photoreceptors, the basis for high-resolution daylight and colour vision, survived and remained functional for extended periods of time. These findings strongly support the hypothesis of deleterious calcium (Ca2+)-influx as the cause of rapid rod cell death and highlight the importance of CNG channels in this process. Furthermore, our findings suggest that targeting rod CNG channels, rather than general Ca2+-channel blockade, is a most promising symptomatic approach to treat otherwise incurable forms of cGMP-related RP.