cGMP Accumulation Causes Photoreceptor Degeneration in CNG Channel Deficiency: Evidence of cGMP Cytotoxicity Independently of Enhanced CNG Channel Function
11-Sep-2013
The Journal of Neuroscience, 2013, doi:10.1523/JNEUROSCI.0909-13.2013, 33(37):14939-14948, published on 11.09.2013
The Journal of Neuroscience, online article
The Journal of Neuroscience, online article
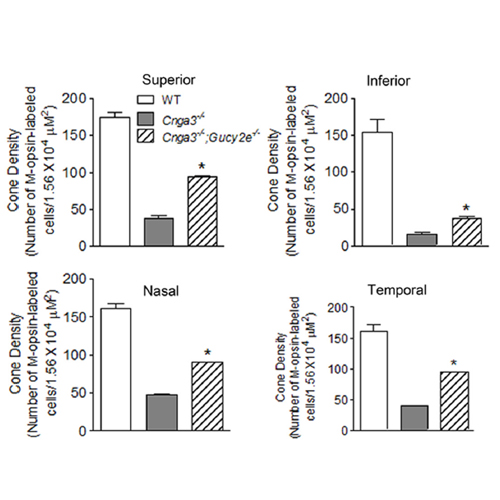
Photoreceptor cyclic nucleotide-gated (CNG) channels regulate Ca2+ influx in rod and cone photoreceptors. cGMP, the native ligand of the photoreceptor CNG channels, has been associated with cytotoxicity when its levels rise above normal due to defects in photoreceptor phosphodiesterase (PDE6) or regulation of retinal guanylyl cyclase (retGC). We found a massive accumulation of cGMP in CNGA3-deficient retina and investigated whether cGMP accumulation plays a role in cone degeneration in CNG channel deficiency. The time course study showed that the retinal cGMP level in Cnga3−/−;Nrl−/− mice with CNGA3 deficiency on a cone-dominant background was sharply increased at postnatal day 8 (P8), peaked around P10–P15, remained high through P30–P60, and returned to near control level at P90. This elevation pattern correlated with photoreceptor apoptotic death, which peaked around P15–P20. In Cnga3−/−;Gucy2e−/− mice lacking retGC1, cone density and expression levels of cone-specific proteins were significantly increased compared with Cnga3−/−, consistent with a role of cGMP accumulation as the major contributor to cone death caused by CNG channel deficiency. The activity and expression levels of cGMP-dependent protein kinase G (PKG) were significantly increased in Cnga3−/−;Nrl−/− retina compared with Nrl−/−, suggesting an involvement of PKG regulation in cell death. Our results indicate that cGMP accumulation in photoreceptors can itself exert cytotoxic effect in cones, independently of CNG channel activity and Ca2+ influx.